Powered Endoscopic Debridement (PED)
EndoRotor® Features:
The EndoRotor PED™ Catheter is the first clincally validated solution for endoscopic debridement of Walled-Off Necrosis (WON) following EUS guided drainage.- Non-thermal mechanical debridement and removal of necrotic tissue in symptomatic WOPN/WON1-9
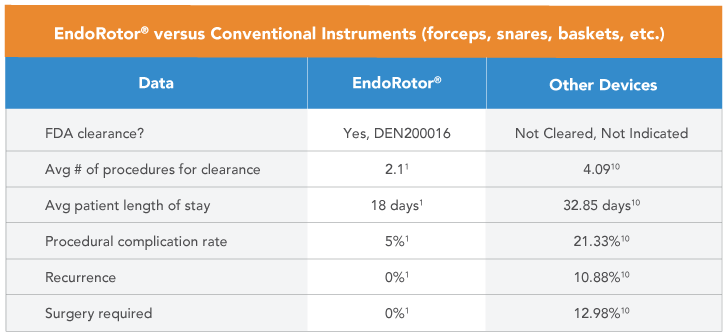
- In multiple clinical trials using EndoRotor® for DEN, there were no SAEs or adverse events (AE) related to the device and a 10% procedure AE rate related to the procedure, demonstrating an improvement in safety over conventional DEN.1-9
- EndoRotor® enables the user to maintain the instrument within the collection which improves procedural efficiency resulting in fewer treatments with reduced risk of stent dislodgement1,4-6
- In a clinical study, patients undergoing treatment of WOPN/WON using EndoRotor® averaged 18 days LOS.1 Whereas patients undergoing treatment for WOPN/WON using conventional, non-FDA cleared instruments averaged 32 days LOS.10, 11
EndoRotor PED™ Safety and Efficacy
Media
Testimonials
FAQs
How does the EndoRotor® work? . . .
The EndoRotor® is an interventional endoscopic tool for debriding tissue. The device is passed directly into the WOPN/WON cavity through a lumen-apposing metal stent and, under direct visualization, tissue is aspirated into the catheter and cut simultaneously. The debrided tissue is then collected in a suction jar.
How does the EndoRotor® help me? What are the advantages over how I perform resection today? . . .
EndoRotor® is the only cleared device for direct endoscopic necrosectomy. Additionally, conventional necrosectomy requires blind manipulation of necrosis, increasing the risk of procedural complications.7 EndoRotor® provides the endoscopist a dedicated and effective tool for the debridement of WOPN/WON under direct visualization without the need to pass the scope in and out of the collection.
What about pathological assessment of the walled-off pancreatic necrosis? . . .
The device collects aspirated material in a vacuum cannister which can be sent for pathological assessment.
What about bleeding? . . .
There was recently a case published in video GIE demonstrating EndoRotor’s® safety and efficacy in debriding a collection containing a visibly exposed superior mesenteric artery.12 In multiple clinical trials using EndoRotor® for DEN, there were no SAEs or adverse events (AE) related to the device and a 10% AE rate related to the procedure. 1-6
Will I need to pass the scope in and out of the collection to remove the debris? . . .
Conventional instruments used for DEN require the user to resect necrotic debris followed by removal of the endoscope through the lumen apposing metal stent (LAMS) to deposit the debris in the stomach which is correlated to an increased number of procedures, increased procedure time, and stent dislodgement.10,11 EndoRotor® enables the user to maintain the instrument within the collection which resulted in fewer treatments and reduced the risk of stent dislodgement. 1,4-6
References
1. Stassen P, et al. Prospective trial evaluating the safety and effectiveness of the EndoRotor® for direct endoscopic necrosectomy of WON (EndoRotor® DEN Trial). Oral Presentation. Digestive Disease Week 2020. doi: 10.1016/j.gie.2020.03.622.
2. Rizzatti G, et al. Endorotor®-based endoscopic necrosectomy avoiding the superior mesenteric artery. Endoscopy. 2020 Apr 24. doi: 10.1055/a-1151-4694.
3. Mangas-Sanjuan C, et al. Endoscopic management of pancreatic necrosis using the EndoRotor®. Endoscopy. 2020;Apr;52(S01):S323. doi: 10.1055/s-0040-1705043.
4. van der Wiel SE, et al. Preliminary report on the safety and utility of a novel automated mechanical endoscopic tissue resection tool for endoscopic necrosectomy: a case series. Endosc Int Open. 2020;Mar;8(3):E274-E280. doi: 10.1055/a-1079-5015.
5. Morris L, et al. EndoRotor® use to manage walled-off pancreatic ne-crosis; first UK experience, Gut. 2019;Jun;68(Suppl 2):A160. doi: 10.1136/gutjnl-2019-BSGAbstracts.302.
6. Schlag C, et al. Endoscopic microdebrider-assisted necrosectomy for walled-off pancreatic necrosis. Endoscopy. 2020;52(S01):S29-S30. doi: 10.1055/s-0040-1704093.
7. Soota K, et al. Experience with endorotor-xt for endoscopic necrosectomy in patients with acute necrotic pancreatitis at a tertiary care center. Gastrointestinal Endos- copy, vol. 91, no. 6, 2020, doi:10.1016/j.gie.2020.03.1133.
8. Mangas-Sanjuan C, et al. Endoscopic management of pancreatic necrosis using the EndoRotor. Endoscopy. 2020;Apr;52(S01):S323. doi: 10.1055/s-0040-1705043.
9. Schlag C, Abdelhafez M, Friedrich-Rust M, Kowalski T, Lorren D, Chiang A, Schlachter- mann A, Siddiqui U, Villa E, Trindade A, Benias P, Hwang JH, Hahid H, Khaleh M, Tyberg A, Koch AD, Bruno MJ. Endoscopic microdebrider-assisted necrosectomy for walled- off pancreatic necrosis. Endoscopy. 2020;52(S01):S29-S30. doi: 10.1055/s-0040- 1704093.
10. Puli SR, et al. Endoscopic transmural necrosectomy for walled-off pancreatic necrosis: a systematic review and meta-analysis. Can j gastroenterol hepatol 2014; 28(1):50-53.
11. Yan L, Dargan A, Nieto J, Shariaha RZ, Binmoeller KF, Adler DG, et al. Direct endoscopic necrosectomy at the time of transmural stent placement results in earlier resolution of complex walled-off pancreatic necrosis: Results from a large multicenter United States trial. Endosc Ultrasound 2019;8:172-9.
12. Rizzatti G, Rimbaș M, De Riso M, Impagnatiello M, Costamagna G, Larghi A. Endorotor-based endoscopic necrosectomy avoiding the superior mesenteric artery. Endoscopy. 2020 Apr 24. doi: 10.1055/a-1151-4694.